Authorized generics offer brand-name drug effectiveness at generic prices. Learn how insurance formularies place them, why they save money, and how to ensure you get the best coverage.
Monoclonal antibody biosimilars offer proven, cost-effective alternatives to expensive biologic drugs like Herceptin and Rituxan. With multiple approved examples and growing clinical use, they're changing cancer and autoimmune care without sacrificing safety.
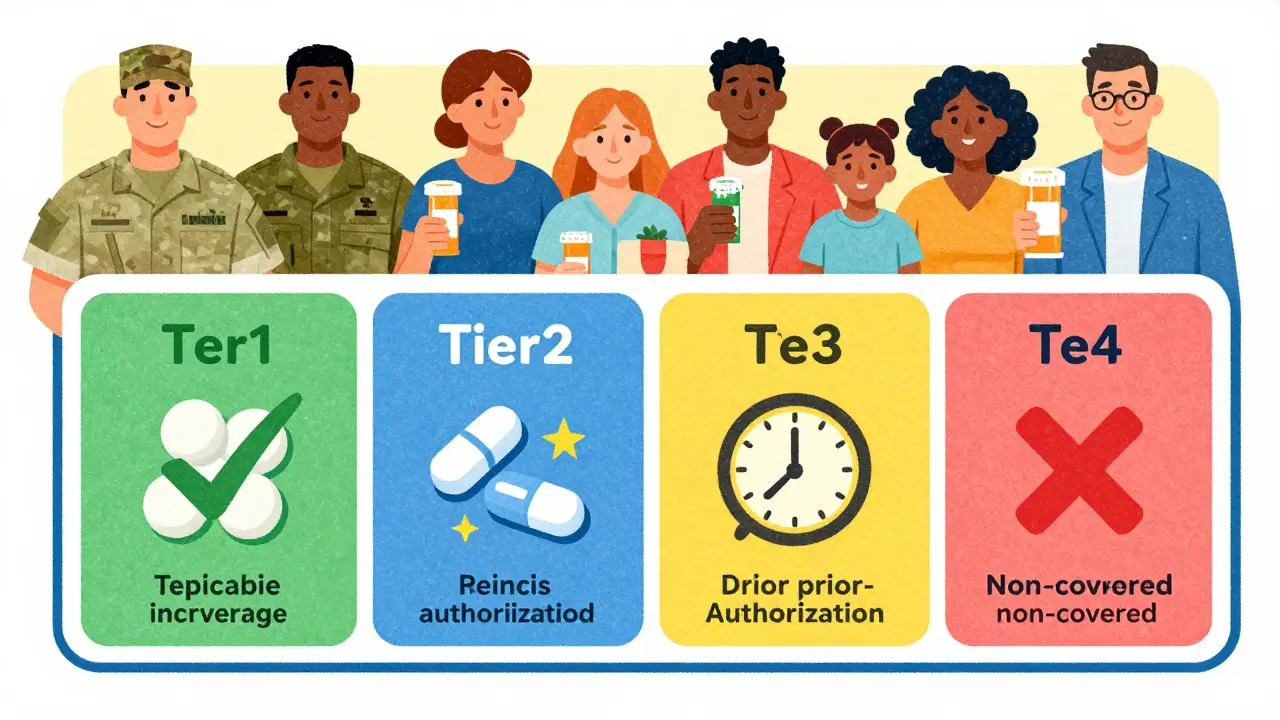
TRICARE covers over 5,500 generic and brand-name drugs. Learn how to check formulary status, compare copays for military pharmacies, home delivery, and retail options, and navigate prior authorizations. Updated for 2026 with latest copay changes.
Learn the early warning signs of dangerous medication side effects that could be life-threatening. Know when to act fast, what symptoms to watch for, and how to protect yourself from serious drug reactions.
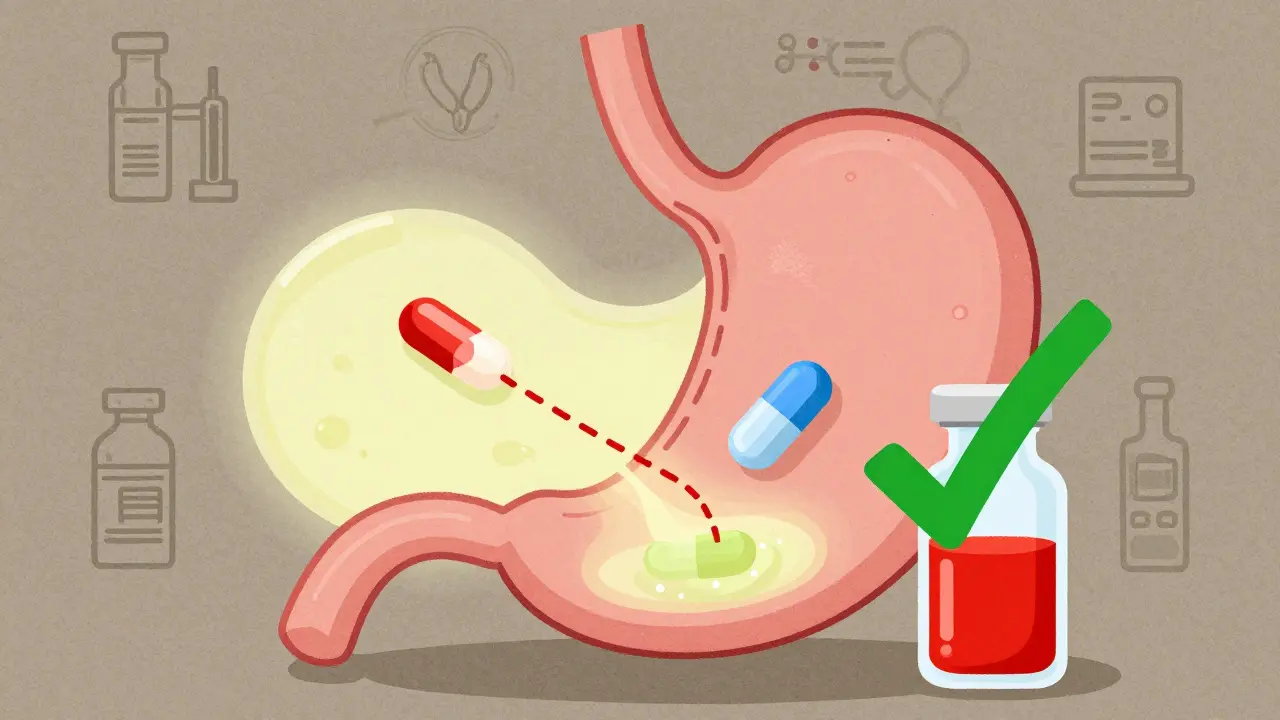
H2 blockers like famotidine and cimetidine can reduce the effectiveness of antivirals and antifungals by raising stomach pH and interfering with liver enzymes. Learn which drugs are at risk and how to avoid dangerous interactions.
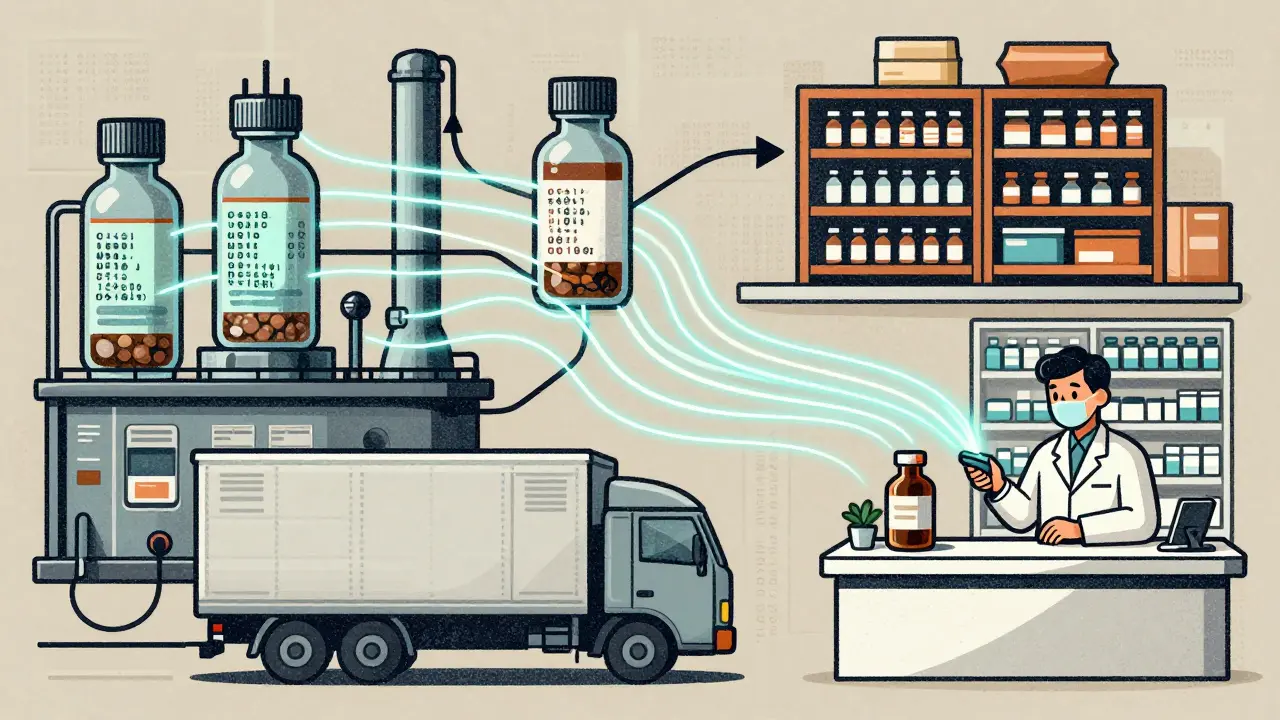
The DSCSA track-and-trace system is the U.S. government's solution to stop counterfeit drugs by requiring every prescription package to have a unique digital identifier. By 2024, all manufacturers, wholesalers, and pharmacies must verify drugs electronically to ensure safety.
Learn how healthcare providers navigate prior authorization to get generic medications approved by insurers, including documentation requirements, submission methods, approval timelines, and practical strategies to reduce delays and denials.
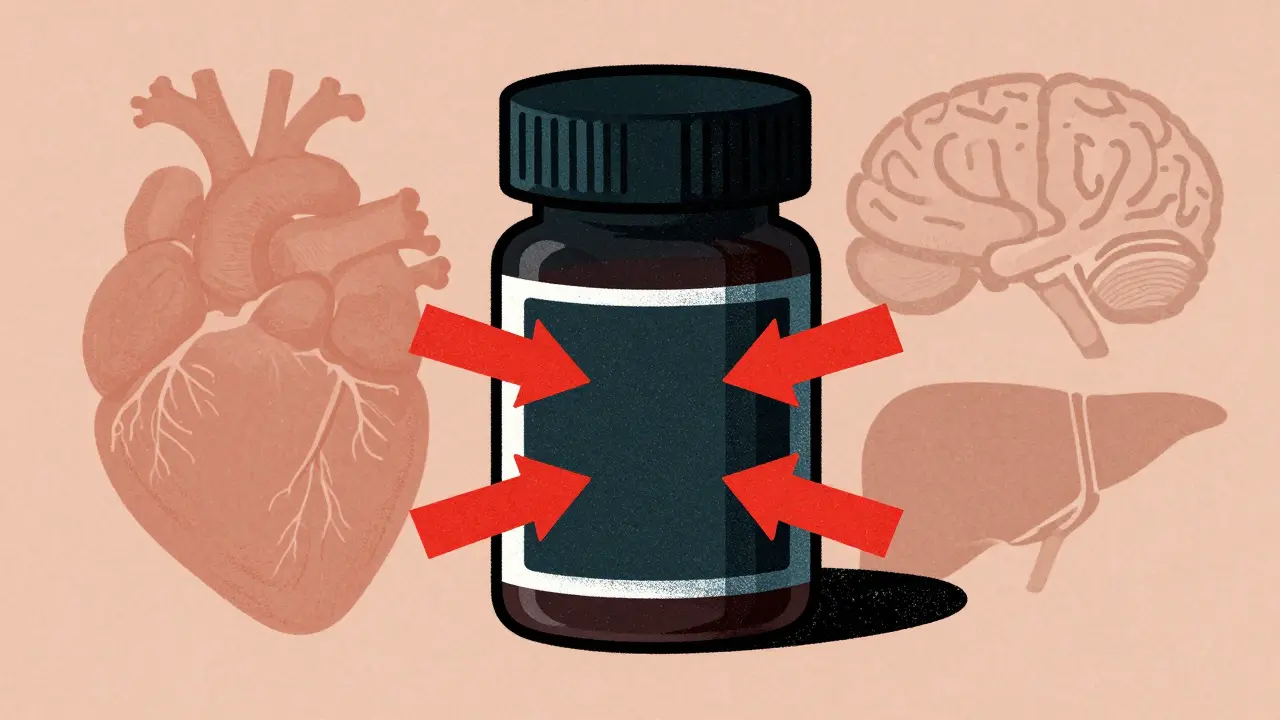
FDA black box warnings are the strongest safety alerts for prescription drugs, signaling life-threatening risks. Learn what they mean, which drugs carry them, and how to stay safe.
Generic drugs are cheaper, but manufacturing defects like capping, contamination, and weight variation are rising. These flaws can lead to underdosing, overdosing, or treatment failure. Here’s what’s wrong-and what’s being done.
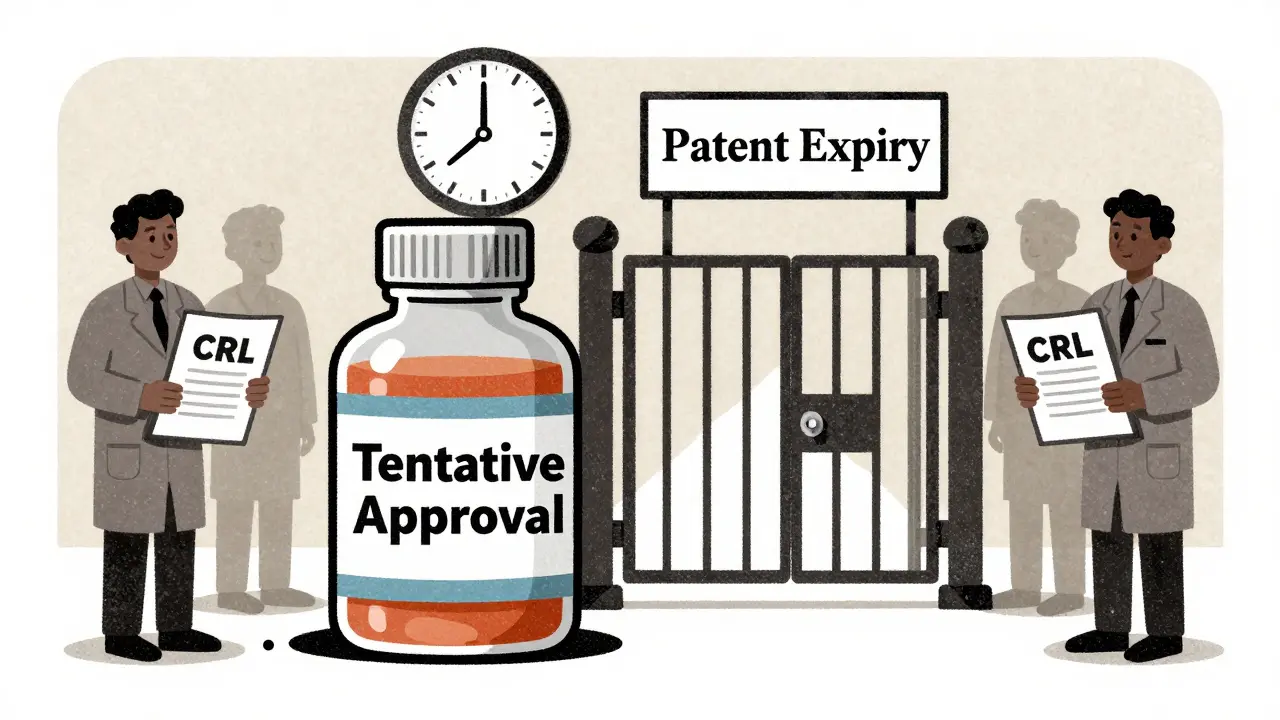
Tentative FDA approval for generics means the drug is scientifically ready - but still can't be sold due to patents, lawsuits, or manufacturing issues. Learn why most generics sit idle for over a year after approval.