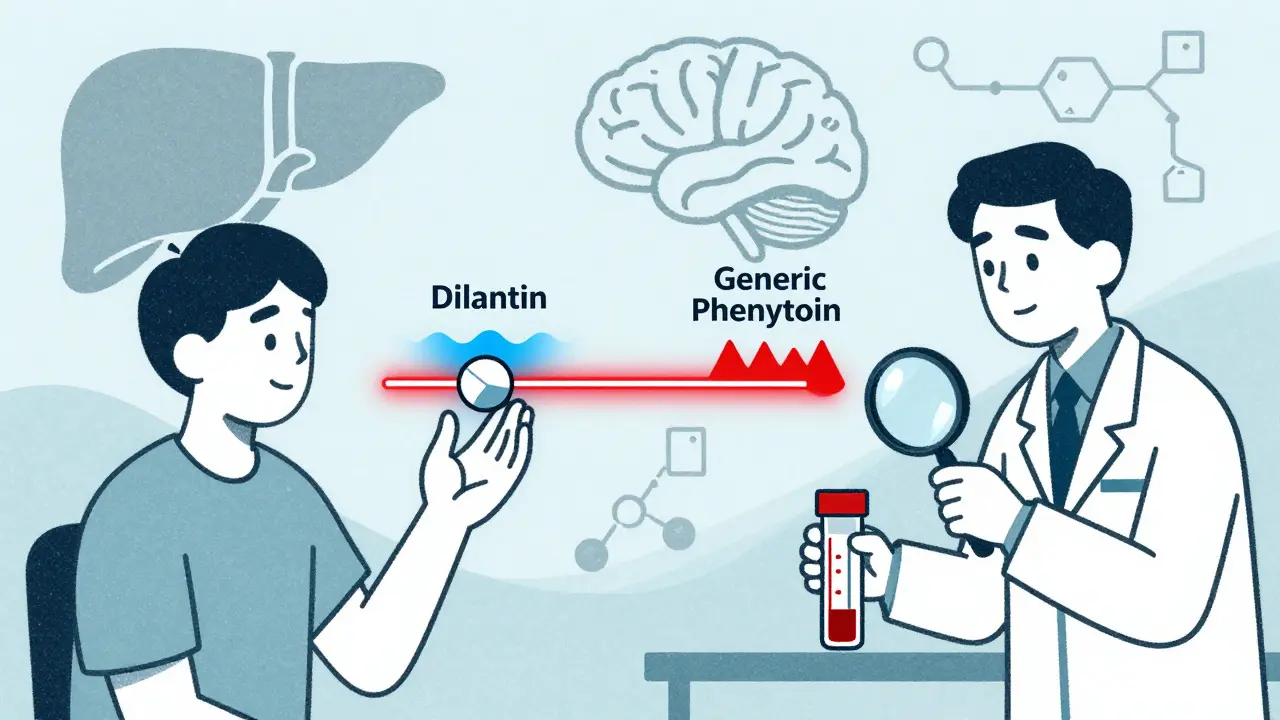
When a doctor prescribes phenytoin for seizures, most patients assume any pill with the same name will work the same way. But with generic versions, that’s not always true. Phenytoin has a narrow therapeutic index - meaning the difference between an effective dose and a toxic one is razor-thin. That’s why switching between brand-name Dilantin and generic phenytoin, or even between different generic brands, can be risky without careful monitoring.
Why Phenytoin Is Different From Other Drugs
Phenytoin doesn’t follow the usual rules of drug dosing. Most medications clear from the body at a steady rate - double the dose, double the concentration. Not phenytoin. At higher levels, its metabolism slows down dramatically. This is called non-linear pharmacokinetics. A small increase in dose - say, 25 to 50 mg - can push levels from safe into toxic territory. One study found that going from 20 mg/L to 25 mg/L could cause severe side effects like ataxia (loss of coordination) and nystagmus (involuntary eye movements). Above 40 mg/L, patients can become confused or unresponsive. Above 100 mg/L, death is possible.Another problem: phenytoin is 90-95% bound to proteins in the blood. Only the small unbound fraction (about 10%) actually works to stop seizures. If protein levels drop - like in malnutrition, liver disease, or kidney failure - more drug floats free. That means even if the total blood level looks normal, the active part might be dangerously high.
Generic Substitutions Aren’t Always Safe
The FDA allows generic drugs to vary by up to 20% in absorption compared to the brand-name version. For most drugs, that’s fine. For phenytoin? Not even close. That 20% variation can swing a patient from seizure-free to seizure-prone - or into toxicity. There are documented cases where patients switched from one generic to another and started having seizures again. Others developed tremors, dizziness, or slurred speech after the switch.Why does this happen? Two reasons. First, different generics use different inactive ingredients (fillers, coatings, binders). These can change how quickly the drug dissolves in the gut. Second, phenytoin’s metabolism depends on liver enzymes (CYP2C9 and CYP2C19). Some generic formulations might interact differently with other medications or even with food. A patient on cimetidine or fluconazole might see levels spike after a switch, even if the dose didn’t change.
When to Check Phenytoin Levels
Routine monitoring isn’t needed for everyone. But if you’re switching formulations - brand to generic, generic to generic, or even switching manufacturers - you need a level check. Here’s when:- Before switching: Get a trough level (just before your next dose) to establish your baseline.
- 5 to 10 days after switching: This is when steady-state levels are reached. Check again.
- After any dose change: Even if you’re staying on the same brand, increasing or decreasing your dose requires a follow-up level.
- If you develop new symptoms: Dizziness, confusion, unsteadiness, or worsening seizures? Get tested.
Don’t check levels too early. Phenytoin takes time to stabilize. Levels drawn within the first 5 days of starting or changing the drug won’t reflect your true steady-state concentration. The NHS Tayside guidelines say to wait at least 5 days - and ideally 10 - after a dose change or formulation switch.
Special Cases: Who Needs Extra Care
Some patients are at higher risk. If you fall into any of these groups, monitoring is non-negotiable:- Older adults: Liver and kidney function decline with age, slowing drug clearance.
- People with low albumin: Low protein levels mean more free phenytoin. Corrected levels can help, but they’re not perfect. Free phenytoin testing is more accurate.
- Patients with liver disease: Phenytoin is metabolized in the liver. Damage here increases toxicity risk.
- Those on other medications: Drugs like amiodarone, valproate, or fluconazole can raise phenytoin levels. Rifampin or carbamazepine can lower them.
- Patients with recent seizures: If seizures return after a switch, check levels immediately.
For patients with low albumin, the correction formula is: Corrected phenytoin = Measured level / ((0.9 × Albumin / 42) + 0.1). But even this isn’t foolproof. Clinical symptoms matter more than numbers.
What to Monitor Beyond Blood Levels
Phenytoin doesn’t just affect your brain. Long-term use can cause serious side effects - regardless of whether you’re on brand or generic:- Gingival hyperplasia: Swollen, overgrown gums. Regular dental checkups are essential.
- Hirsutism and facial changes: Increased body hair and coarsening of facial features.
- Bone problems: Phenytoin reduces vitamin D, leading to low calcium, low phosphate, and weakened bones. Get bone density and vitamin D levels checked every 2-5 years.
- Blood cell changes: Rarely, it can cause low white cells or platelets. A CBC every 6-12 months is recommended.
- Liver function: Monitor enzymes (ALT, AST) annually.
These issues happen over time. They’re not caused by switching generics - but they’re still part of your overall care. Don’t ignore them just because your seizure control seems fine.
What Patients Should Do
If you take phenytoin:- Ask your pharmacist: Is this the same brand I’ve been taking?
- Don’t accept a refill if the pill looks different - even if the name is the same.
- Report any new symptoms - even if they seem minor.
- Keep a log: Date of switch, dose, symptoms, and when you had your last level.
- Insist on a level check after any formulation change.
Phenytoin isn’t like taking aspirin. It’s a drug that demands attention. A single switch can undo months of stable control. That’s why therapeutic drug monitoring isn’t optional - it’s a safety net.
Dan Mayer
March 10, 2026 AT 03:59
lol so generics are bad? guess i'll just pay $500 a month for dilantin then. my cat needs phenytoin too and she's not paying for brand name. #firstworldproblems
Neeti Rustagi
March 10, 2026 AT 12:35
The pharmacokinetic variability of phenytoin is a well-documented clinical challenge, particularly in resource-constrained settings where generic substitution is unavoidable. The non-linear metabolism necessitates individualized therapeutic drug monitoring, and reliance on total serum concentrations without accounting for albumin-binding alterations can lead to significant clinical misinterpretation. In India, where polypharmacy and malnutrition are common, free phenytoin levels should be prioritized over total levels when available.
Janelle Pearl
March 12, 2026 AT 06:41
I had a patient last month switch generics and start having seizures again. She was terrified. We checked her level-dropped from 18 to 11. Took 3 weeks to stabilize. No one told her the pill looked different was a big deal. Doctors forget how scary this stuff is. 😔
Katy Shamitz
March 12, 2026 AT 14:49
People who don't get their levels checked after switching generics are basically playing Russian roulette with their brain. If you're too cheap to monitor, maybe you shouldn't be on a drug that can kill you. Just sayin'.
Nicholas Gama
March 13, 2026 AT 15:52
FDA doesn't care. Big Pharma owns them. You think this is an accident? Nah. It's profit. 20% variance? That's a backdoor for generics to kill people quietly. I've seen the memos.
Mary Beth Brook
March 15, 2026 AT 12:12
American patients need to stop accepting subpar generics. We have the best pharma in the world. Why settle for foreign-manufactured fillers that don't dissolve right? Dilantin is the gold standard. Period.
Ray Foret Jr.
March 16, 2026 AT 11:23
I’ve been on phenytoin for 12 years. Switched generics 3 times. Never had an issue. But I do check my levels every 6 months like my neuro said. 🙌 Maybe not everyone’s a disaster waiting to happen?
Samantha Fierro
March 18, 2026 AT 09:41
Thank you for writing this. So many patients feel powerless when their meds change without warning. The emotional toll is real-fear, confusion, loss of control. Clinicians need to sit down, explain the risks plainly, and validate those fears. A simple ‘I know this is scary’ goes further than you think.
Robert Bliss
March 20, 2026 AT 03:41
I get why generics are cheaper. But if a drug can make you dizzy or stop working, maybe we need better standards. Not everyone can afford blood tests. Maybe we should just make phenytoin a special case? 🤔